Page 24 - Chemistry_Form_2
P. 24
Chemistry
for Secondary Schools
Exercise 1.2
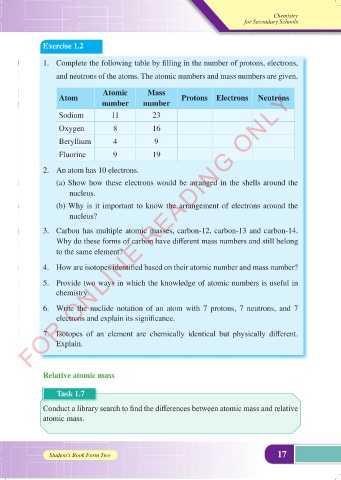
1. Complete the following table by filling in the number of protons, electrons,
and neutrons of the atoms. The atomic numbers and mass numbers are given.
FOR ONLINE READING ONLY
Atomic Mass
Atom Protons Electrons Neutrons
number number
Sodium 11 23
Oxygen 8 16
Beryllium 4 9
Fluorine 9 19
2. An atom has 10 electrons.
(a) Show how these electrons would be arranged in the shells around the
nucleus.
(b) Why is it important to know the arrangement of electrons around the
nucleus?
3. Carbon has multiple atomic masses, carbon-12, carbon-13 and carbon-14.
Why do these forms of carbon have different mass numbers and still belong
to the same element?
4. How are isotopes identified based on their atomic number and mass number?
5. Provide two ways in which the knowledge of atomic numbers is useful in
chemistry.
6. Write the nuclide notation of an atom with 7 protons, 7 neutrons, and 7
electrons and explain its significance.
7. Isotopes of an element are chemically identical but physically different.
Explain.
Relative atomic mass
Task 1.7
Conduct a library search to find the differences between atomic mass and relative
atomic mass.
Student’s Book Form Two 17
01/08/2025 11:20:51
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 17 01/08/2025 11:20:51
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 17