Page 22 - Chemistry_Form_2
P. 22
Chemistry
for Secondary Schools
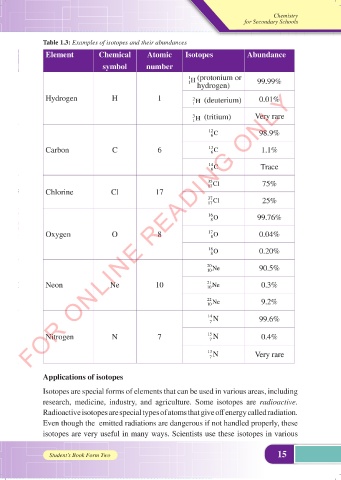
Table 1.3: Examples of isotopes and their abundances
Element Chemical Atomic Isotopes Abundance
symbol number
1 (protonium or
H
1 hydrogen) 99.99%
FOR ONLINE READING ONLY
Hydrogen H 1 H (deuterium) 0.01%
2
1
H (tritium) Very rare
3
1
12 C 98.9%
6
Carbon C 6 13 6 C 1.1%
14 C Trace
6
35 Cl 75%
17
Chlorine Cl 17
37 Cl 25%
17
16 O 99.76%
8
Oxygen O 8 17 8 O 0.04%
18 O 0.20%
8
20 Ne 90.5%
10
Neon Ne 10 21 Ne 0.3%
10
22 Ne 9.2%
10
14 N 99.6%
7
Nitrogen N 7 15 7 N 0.4%
13 N Very rare
7
Applications of isotopes
Isotopes are special forms of elements that can be used in various areas, including
research, medicine, industry, and agriculture. Some isotopes are radioactive.
Radioactive isotopes are special types of atoms that give off energy called radiation.
Even though the emitted radiations are dangerous if not handled properly, these
isotopes are very useful in many ways. Scientists use these isotopes in various
Student’s Book Form Two 15
01/08/2025 11:20:51
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 15 01/08/2025 11:20:51
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 15