Page 76 - Chemistry_Form_2
P. 76
Chemistry
for Secondary Schools
Nomenclature of binary covalent compounds
Covalent compounds are formed between two non-metal elements. These
compounds are named differently from ionic compounds. The number of atoms
are presented by prefixes as shown in Table 3.4.
FOR ONLINE READING ONLY
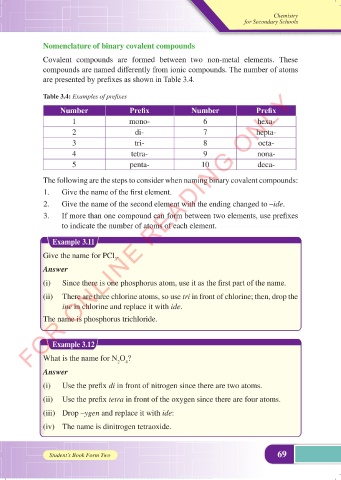
Table 3.4: Examples of prefixes
Number Prefix Number Prefix
1 mono- 6 hexa-
2 di- 7 hepta-
3 tri- 8 octa-
4 tetra- 9 nona-
5 penta- 10 deca-
The following are the steps to consider when naming binary covalent compounds:
1. Give the name of the first element.
2. Give the name of the second element with the ending changed to –ide.
3. If more than one compound can form between two elements, use prefixes
to indicate the number of atoms of each element.
Example 3.11
Give the name for PCl .
3
Answer
(i) Since there is one phosphorus atom, use it as the first part of the name.
(ii) There are three chlorine atoms, so use tri in front of chlorine; then, drop the
ine in chlorine and replace it with ide.
The name is phosphorus trichloride.
Example 3.12
What is the name for N O ?
2 4
Answer
(i) Use the prefix di in front of nitrogen since there are two atoms.
(ii) Use the prefix tetra in front of the oxygen since there are four atoms.
(iii) Drop –ygen and replace it with ide:
(iv) The name is dinitrogen tetraoxide.
Student’s Book Form Two 69
01/08/2025 11:20:57
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 69 01/08/2025 11:20:57
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 69