Page 77 - Chemistry_Form_2
P. 77
Chemistry
for Secondary Schools
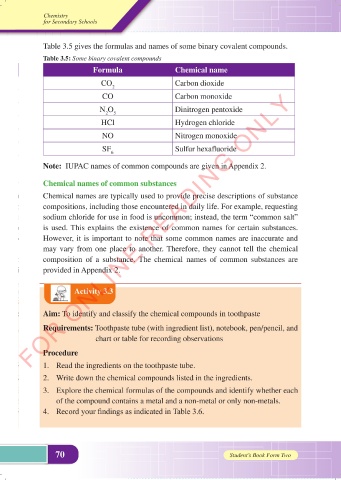
Table 3.5 gives the formulas and names of some binary covalent compounds.
Table 3.5: Some binary covalent compounds
Formula Chemical name
CO 2 Carbon dioxide
FOR ONLINE READING ONLY
CO Carbon monoxide
N O Dinitrogen pentoxide
2 5
HCl Hydrogen chloride
NO Nitrogen monoxide
SF 6 Sulfur hexafluoride
Note: IUPAC names of common compounds are given in Appendix 2.
Chemical names of common substances
Chemical names are typically used to provide precise descriptions of substance
compositions, including those encountered in daily life. For example, requesting
sodium chloride for use in food is uncommon; instead, the term “common salt”
is used. This explains the existence of common names for certain substances.
However, it is important to note that some common names are inaccurate and
may vary from one place to another. Therefore, they cannot tell the chemical
composition of a substance. The chemical names of common substances are
provided in Appendix 2.
Activity 3.3
Aim: To identify and classify the chemical compounds in toothpaste
Requirements: Toothpaste tube (with ingredient list), notebook, pen/pencil, and
chart or table for recording observations
Procedure
1. Read the ingredients on the toothpaste tube.
2. Write down the chemical compounds listed in the ingredients.
3. Explore the chemical formulas of the compounds and identify whether each
of the compound contains a metal and a non-metal or only non-metals.
4. Record your findings as indicated in Table 3.6.
70 Student’s Book Form Two
01/08/2025 11:20:57
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 70 01/08/2025 11:20:57
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 70