Page 129 - Chemistry_Form_2
P. 129
Chemistry
for Secondary Schools
Questions
1. What colour changes were observed using cassava leaf extract indicator in the
substances tested?
2. How can the knowledge gained from this activity be used in testing spoiled
FOR ONLINE READING ONLY
fruits?
Synthetic pH indicators
Synthetic pH indicators are chemical substances which are made from different
substances in the laboratories used to determine the pH of substances. Examples
of synthetic indicators include universal indicator, phenolphthalein (POP), and
methyl orange (MO).
Universal indicator
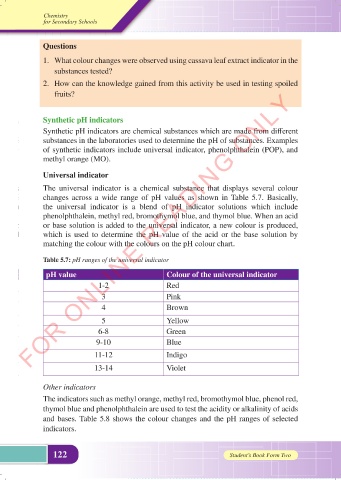
The universal indicator is a chemical substance that displays several colour
changes across a wide range of pH values as shown in Table 5.7. Basically,
the universal indicator is a blend of pH indicator solutions which include
phenolphthalein, methyl red, bromothymol blue, and thymol blue. When an acid
or base solution is added to the universal indicator, a new colour is produced,
which is used to determine the pH value of the acid or the base solution by
matching the colour with the colours on the pH colour chart.
Table 5.7: pH ranges of the universal indicator
pH value Colour of the universal indicator
1-2 Red
3 Pink
4 Brown
5 Yellow
6-8 Green
9-10 Blue
11-12 Indigo
13-14 Violet
Other indicators
The indicators such as methyl orange, methyl red, bromothymol blue, phenol red,
thymol blue and phenolphthalein are used to test the acidity or alkalinity of acids
and bases. Table 5.8 shows the colour changes and the pH ranges of selected
indicators.
122 Student’s Book Form Two
01/08/2025 11:21:07
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 122
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 122 01/08/2025 11:21:07