Page 125 - Chemistry_Form_2
P. 125
Chemistry
for Secondary Schools
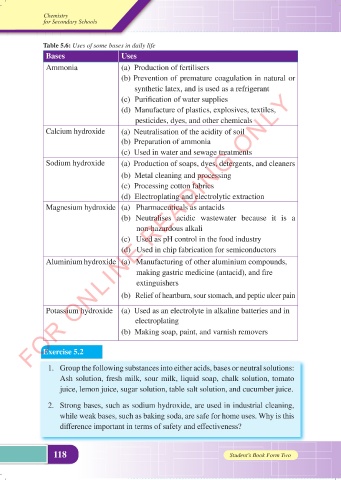
Table 5.6: Uses of some bases in daily life
Bases Uses
Ammonia (a) Production of fertilisers
(b) Prevention of premature coagulation in natural or
synthetic latex, and is used as a refrigerant
FOR ONLINE READING ONLY
(c) Purification of water supplies
(d) Manufacture of plastics, explosives, textiles,
pesticides, dyes, and other chemicals
Calcium hydroxide (a) Neutralisation of the acidity of soil
(b) Preparation of ammonia
(c) Used in water and sewage treatments
Sodium hydroxide (a) Production of soaps, dyes, detergents, and cleaners
(b) Metal cleaning and processing
(c) Processing cotton fabrics
(d) Electroplating and electrolytic extraction
Magnesium hydroxide (a) Pharmaceuticals as antacids
(b) Neutralises acidic wastewater because it is a
non-hazardous alkali
(c) Used as pH control in the food industry
(d) Used in chip fabrication for semiconductors
Aluminium hydroxide (a) Manufacturing of other aluminium compounds,
making gastric medicine (antacid), and fire
extinguishers
(b) Relief of heartburn, sour stomach, and peptic ulcer pain
Potassium hydroxide (a) Used as an electrolyte in alkaline batteries and in
electroplating
(b) Making soap, paint, and varnish removers
Exercise 5.2
1. Group the following substances into either acids, bases or neutral solutions:
Ash solution, fresh milk, sour milk, liquid soap, chalk solution, tomato
juice, lemon juice, sugar solution, table salt solution, and cucumber juice.
2. Strong bases, such as sodium hydroxide, are used in industrial cleaning,
while weak bases, such as baking soda, are safe for home uses. Why is this
difference important in terms of safety and effectiveness?
118 Student’s Book Form Two
01/08/2025 11:21:06
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 118 01/08/2025 11:21:06
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 118