Page 130 - Chemistry_Form_2
P. 130
Chemistry
for Secondary Schools
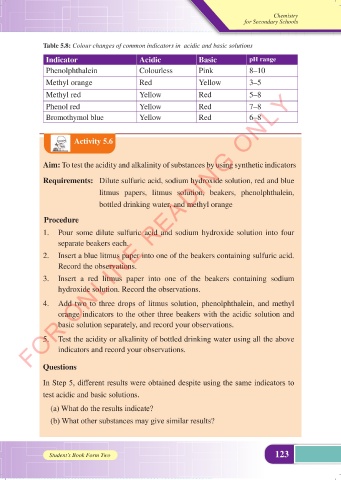
Table 5.8: Colour changes of common indicators in acidic and basic solutions
Indicator Acidic Basic pH range
Phenolphthalein Colourless Pink 8–10
Methyl orange Red Yellow 3–5
FOR ONLINE READING ONLY
Methyl red Yellow Red 5–8
Phenol red Yellow Red 7–8
Bromothymol blue Yellow Red 6–8
Activity 5.6
Aim: To test the acidity and alkalinity of substances by using synthetic indicators
Requirements: Dilute sulfuric acid, sodium hydroxide solution, red and blue
litmus papers, litmus solution, beakers, phenolphthalein,
bottled drinking water, and methyl orange
Procedure
1. Pour some dilute sulfuric acid and sodium hydroxide solution into four
separate beakers each.
2. Insert a blue litmus paper into one of the beakers containing sulfuric acid.
Record the observations.
3. Insert a red litmus paper into one of the beakers containing sodium
hydroxide solution. Record the observations.
4. Add two to three drops of litmus solution, phenolphthalein, and methyl
orange indicators to the other three beakers with the acidic solution and
basic solution separately, and record your observations.
5. Test the acidity or alkalinity of bottled drinking water using all the above
indicators and record your observations.
Questions
In Step 5, different results were obtained despite using the same indicators to
test acidic and basic solutions.
(a) What do the results indicate?
(b) What other substances may give similar results?
Student’s Book Form Two 123
01/08/2025 11:21:07
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 123 01/08/2025 11:21:07
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 123