Page 16 - Chemistry_Form_2
P. 16
Chemistry
for Secondary Schools
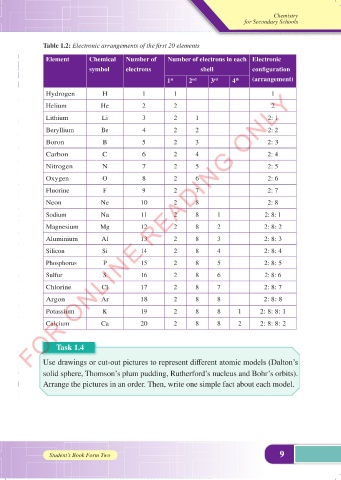
Table 1.2: Electronic arrangements of the first 20 elements
Element Chemical Number of Number of electrons in each Electronic
symbol electrons shell configuration
1 2 3 4 (arrangement)
st
rd
th
nd
FOR ONLINE READING ONLY
Hydrogen H 1 1 1
Helium He 2 2 2
Lithium Li 3 2 1 2: 1
Beryllium Be 4 2 2 2: 2
Boron B 5 2 3 2: 3
Carbon C 6 2 4 2: 4
Nitrogen N 7 2 5 2: 5
Oxygen O 8 2 6 2: 6
Fluorine F 9 2 7 2: 7
Neon Ne 10 2 8 2: 8
Sodium Na 11 2 8 1 2: 8: 1
Magnesium Mg 12 2 8 2 2: 8: 2
Aluminium Al 13 2 8 3 2: 8: 3
Silicon Si 14 2 8 4 2: 8: 4
Phosphorus P 15 2 8 5 2: 8: 5
Sulfur S 16 2 8 6 2: 8: 6
Chlorine Cl 17 2 8 7 2: 8: 7
Argon Ar 18 2 8 8 2: 8: 8
Potassium K 19 2 8 8 1 2: 8: 8: 1
Calcium Ca 20 2 8 8 2 2: 8: 8: 2
Task 1.4
Use drawings or cut-out pictures to represent different atomic models (Dalton’s
solid sphere, Thomson’s plum pudding, Rutherford’s nucleus and Bohr’s orbits).
Arrange the pictures in an order. Then, write one simple fact about each model.
Student’s Book Form Two 9
01/08/2025 11:20:50
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 9 01/08/2025 11:20:50
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 9