Page 44 - Chemistry_Form_2
P. 44
Chemistry
for Secondary Schools
Procedure
1. Fill three beakers halfway with water.
2. Add a few drops of phenolphthalein or universal indicator to each beaker.
3. Using forceps, carefully drop a small piece of lithium into the first beaker and
FOR ONLINE READING ONLY
observe the reaction.
4. Repeat Step 3 for sodium and potassium in separate beakers.
5. Record observations for each metal, focusing on the rate of reaction, colour
change (due to the indicator), production of gas and sound.
6. Clean up all the apparatus safely under the supervision of the teacher.
Questions
1. Which element was the fastest to react with water?
2. How did the colour of water change and what did it indicate?
3. What trend did you observe in the reactivity of those elements down the
group?
4. How does this experiment support the idea of periodic classification?
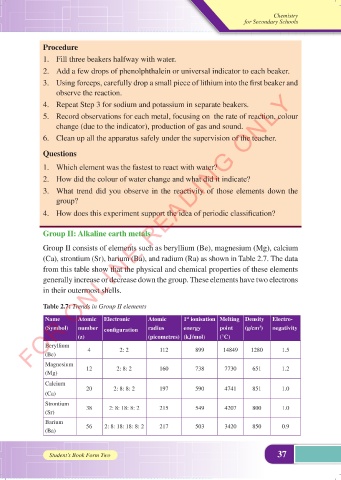
Group II: Alkaline earth metals
Group II consists of elements such as beryllium (Be), magnesium (Mg), calcium
(Ca), strontium (Sr), barium (Ba), and radium (Ra) as shown in Table 2.7. The data
from this table show that the physical and chemical properties of these elements
generally increase or decrease down the group. These elements have two electrons
in their outermost shells.
Table 2.7: Trends in Group II elements
Name Atomic Electronic Atomic 1 ionisation Melting Density Electro-
st
(Symbol) number configuration radius energy point (g/cm ) negativity
3
(z) (picometres) (kJ/mol) (°C)
Beryllium
(Be) 4 2: 2 112 899 14849 1280 1.5
Magnesium 12 2: 8: 2 160 738 7730 651 1.2
(Mg)
Calcium 20 2: 8: 8: 2 197 590 4741 851 1.0
(Ca)
Strontium
(Sr) 38 2: 8: 18: 8: 2 215 549 4207 800 1.0
Barium 56 2: 8: 18: 18: 8: 2 217 503 3420 850 0.9
(Ba)
Student’s Book Form Two 37
01/08/2025 11:20:53
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 37 01/08/2025 11:20:53
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 37