Page 63 - Chemistry_Form_2
P. 63
Chemistry
for Secondary Schools
A radical is a group of atoms which behaves as a single unit and has a positive or
negative charge. It contains at least one unpaired electron. Such a group maintains
its identity throughout any chemical reaction. Most radicals form the non-metallic
part of a compound, so their ions are negatively charged. Examples are CO and
2–
3
SO 4 2– ions. An exception is for the ammonium radical, NH , which behaves like
+
FOR ONLINE READING ONLY
4
the metallic part of a compound and forms a positive ion. The valency of the
radical is the same as the numerical value that the group acquires when it loses
or gains an electron to form an ion. The common radicals with their formulas and
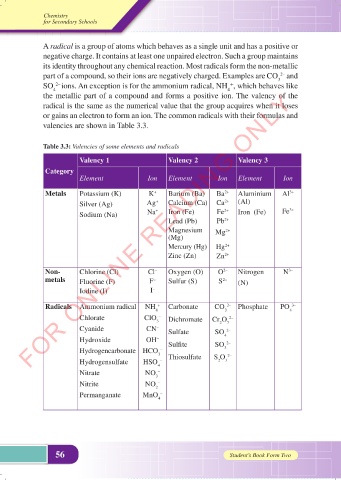
valencies are shown in Table 3.3.
Table 3.3: Valencies of some elements and radicals
Valency 1 Valency 2 Valency 3
Category
Element Ion Element Ion Element Ion
Metals Potassium (K) K + Barium (Ba) Ba 2+ Aluminium Al 3+
Silver (Ag) Ag + Calcium (Ca) Ca 2+ (Al)
Sodium (Na) Na + Iron (Fe) Fe 2+ Iron (Fe) Fe 3+
Lead (Pb) Pb 2+
Magnesium Mg 2+
(Mg)
Mercury (Hg) Hg 2+
Zinc (Zn) Zn 2+
Non- Chlorine (Cl) Cl – Oxygen (O) O 2– Nitrogen N 3–
metals Fluorine (F) F – Sulfur (S) S 2– (N)
Iodine (I) I –
Radicals Ammonium radical NH + Carbonate CO 2– Phosphate PO 3–
4 3 4
Chlorate ClO 3 – Dichromate Cr O 2–
2
7
Cyanide CN – Sulfate SO 2–
Hydroxide OH – Sulfite SO 4 2–
Hydrogencarbonate HCO 3 – 3 2–
Hydrogensulfate HSO – Thiosulfate S O 3
2
4
Nitrate NO –
3
Nitrite NO 2 –
Permanganate MnO –
4
56 Student’s Book Form Two
01/08/2025 11:20:55
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 56 01/08/2025 11:20:55
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 56