Page 91 - Physics_Form_2
P. 91
Current electricity
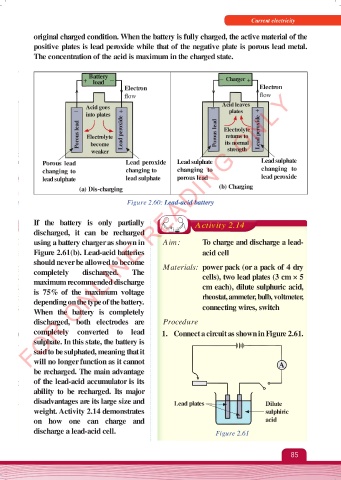
original charged condition. When the battery is fully charged, the active material of the
positive plates is lead peroxide while that of the negative plate is porous lead metal.
The concentration of the acid is maximum in the charged state.
Battery
+ load – – Charger +
Electron Electron
FOR ONLINE READING ONLY
flow flow
– Acid goes + – Acid leaves +
plates
into plates
Porous lead Electrolyte Lead peroxide Porous lead Electrolyte Lead peroxide
returns to
its normal
become
weaker strength
Porous lead Lead peroxide Lead sulphate Lead sulphate
changing to changing to changing to changing to
lead sulphate lead sulphate porous lead lead peroxide
(a) Dis-charging (b) Charging
Figure 2.60: Lead-acid battery
If the battery is only partially Activity 2.14
discharged, it can be recharged
using a battery charger as shown in Aim: To charge and discharge a lead-
Figure 2.61(b). Lead-acid batteries acid cell
should never be allowed to become Materials: power pack (or a pack of 4 dry
completely discharged. The cells), two lead plates (3 cm × 5
maximum recommended discharge cm each), dilute sulphuric acid,
is 75% of the maximum voltage rheostat, ammeter, bulb, voltmeter,
depending on the type of the battery.
When the battery is completely connecting wires, switch
discharged, both electrodes are Procedure
completely converted to lead 1. Connect a circuit as shown in Figure 2.61.
sulphate. In this state, the battery is
said to be sulphated, meaning that it
will no longer function as it cannot A
be recharged. The main advantage
of the lead-acid accumulator is its
ability to be recharged. Its major
disadvantages are its large size and Lead plates Dilute
weight. Activity 2.14 demonstrates sulphiric
on how one can charge and acid
discharge a lead-acid cell. Figure 2.61
85
Physics Form 2 Final.indd 85 25/10/2025 10:26