Page 30 - Chemistry_Form_2
P. 30
Chemistry
for Secondary Schools
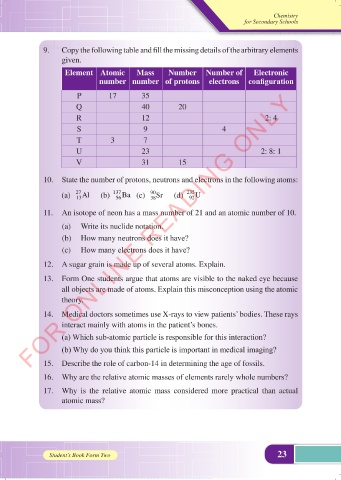
9. Copy the following table and fill the missing details of the arbitrary elements
given.
Element Atomic Mass Number Number of Electronic
number number of protons electrons configuration
FOR ONLINE READING ONLY
P 17 35
Q 40 20
R 12 2: 4
S 9 4
T 3 7
U 23 2: 8: 1
V 31 15
10. State the number of protons, neutrons and electrons in the following atoms:
90
(a) 27 Al (b) 137 Ba (c) Sr (d) 235 U
92
13
38
56
11. An isotope of neon has a mass number of 21 and an atomic number of 10.
(a) Write its nuclide notation.
(b) How many neutrons does it have?
(c) How many electrons does it have?
12. A sugar grain is made up of several atoms. Explain.
13. Form One students argue that atoms are visible to the naked eye because
all objects are made of atoms. Explain this misconception using the atomic
theory.
14. Medical doctors sometimes use X-rays to view patients’ bodies. These rays
interact mainly with atoms in the patient’s bones.
(a) Which sub-atomic particle is responsible for this interaction?
(b) Why do you think this particle is important in medical imaging?
15. Describe the role of carbon-14 in determining the age of fossils.
16. Why are the relative atomic masses of elements rarely whole numbers?
17. Why is the relative atomic mass considered more practical than actual
atomic mass?
Student’s Book Form Two 23
01/08/2025 11:20:52
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 23 01/08/2025 11:20:52
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 23