Page 33 - Chemistry_Form_2
P. 33
Chemistry
for Secondary Schools
Law, which was based on atomic masses, the modern periodic table is structured
according to atomic numbers. Therefore, it led to the development of the modern
Periodic Law which states that the properties of elements are a periodic function
of their atomic numbers. This means that elements exhibit recurring (periodic)
chemical properties when arranged in increasing atomic number, a phenomenon
FOR ONLINE READING ONLY
known as periodicity. For example, lithium, a very reactive metal, has one
electron in its outermost shell. The eighth element after lithium is sodium, which
shares many of the same characteristics as lithium, including a single electron in
its outermost shell and high reactivity. Similarly, the eighth element after sodium
is potassium, which also exhibits the same properties. This periodic recurrence of
chemical behaviour is among the fundamental principles of the modern periodic
table.
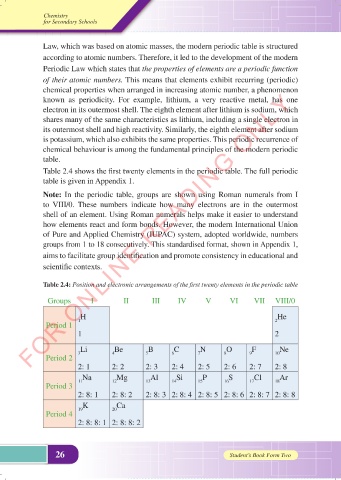
Table 2.4 shows the first twenty elements in the periodic table. The full periodic
table is given in Appendix 1.
Note: In the periodic table, groups are shown using Roman numerals from I
to VIII/0. These numbers indicate how many electrons are in the outermost
shell of an element. Using Roman numerals helps make it easier to understand
how elements react and form bonds. However, the modern International Union
of Pure and Applied Chemistry (IUPAC) system, adopted worldwide, numbers
groups from 1 to 18 consecutively. This standardised format, shown in Appendix 1,
aims to facilitate group identification and promote consistency in educational and
scientific contexts.
Table 2.4: Position and electronic arrangements of the first twenty elements in the periodic table
Groups I II III IV V VI VII VIII/0
H He
Period 1 1 2
1 2
Li Be B C N O F Ne
3 4 5 6 7 8 9 10
Period 2
2: 1 2: 2 2: 3 2: 4 2: 5 2: 6 2: 7 2: 8
Na Mg Al Si P S Cl Ar
11 12 13 14 15 16 17 18
Period 3
2: 8: 1 2: 8: 2 2: 8: 3 2: 8: 4 2: 8: 5 2: 8: 6 2: 8: 7 2: 8: 8
K Ca
Period 4 19 20
2: 8: 8: 1 2: 8: 8: 2
26 Student’s Book Form Two
01/08/2025 11:20:52
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 26
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 26 01/08/2025 11:20:52