Page 36 - Chemistry_Form_2
P. 36
Chemistry
for Secondary Schools
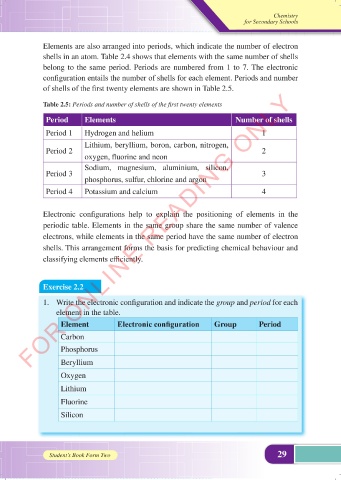
Elements are also arranged into periods, which indicate the number of electron
shells in an atom. Table 2.4 shows that elements with the same number of shells
belong to the same period. Periods are numbered from 1 to 7. The electronic
configuration entails the number of shells for each element. Periods and number
of shells of the first twenty elements are shown in Table 2.5.
FOR ONLINE READING ONLY
Table 2.5: Periods and number of shells of the first twenty elements
Period Elements Number of shells
Period 1 Hydrogen and helium 1
Lithium, beryllium, boron, carbon, nitrogen,
Period 2 2
oxygen, fluorine and neon
Sodium, magnesium, aluminium, silicon,
Period 3 3
phosphorus, sulfur, chlorine and argon
Period 4 Potassium and calcium 4
Electronic configurations help to explain the positioning of elements in the
periodic table. Elements in the same group share the same number of valence
electrons, while elements in the same period have the same number of electron
shells. This arrangement forms the basis for predicting chemical behaviour and
classifying elements efficiently.
Exercise 2.2
1. Write the electronic configuration and indicate the group and period for each
element in the table.
Element Electronic configuration Group Period
Carbon
Phosphorus
Beryllium
Oxygen
Lithium
Fluorine
Silicon
Student’s Book Form Two 29
01/08/2025 11:20:53
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 29 01/08/2025 11:20:53
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 29