Page 9 - Physics_Form_2
P. 9
Static electricity
3. Slowly separate the two sheet. Meanwhile, the protons in the lower plastic
plastic sheets and listen to sheet attract the electrons from the upper sheet.
what occurs. This interaction explains why the sheets attract
4. Bring the plastic sheets closer each other after being rubbed together.
and observe the reaction.
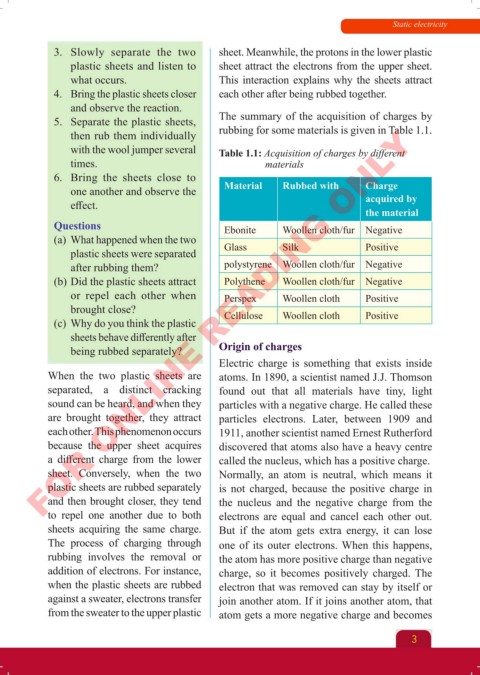
5. Separate the plastic sheets, The summary of the acquisition of charges by
FOR ONLINE READING ONLY
then rub them individually rubbing for some materials is given in Table 1.1.
with the wool jumper several Table 1.1: Acquisition of charges by different
times. materials
6. Bring the sheets close to
one another and observe the Material Rubbed with Charge
effect. acquired by
the material
Questions Ebonite Woollen cloth/fur Negative
(a) What happened when the two
plastic sheets were separated Glass Silk Positive
after rubbing them? polystyrene Woollen cloth/fur Negative
(b) Did the plastic sheets attract Polythene Woollen cloth/fur Negative
or repel each other when Perspex Woollen cloth Positive
brought close? Cellulose Woollen cloth Positive
(c) Why do you think the plastic
sheets behave differently after
being rubbed separately? Origin of charges
Electric charge is something that exists inside
When the two plastic sheets are atoms. In 1890, a scientist named J.J. Thomson
separated, a distinct cracking found out that all materials have tiny, light
sound can be heard, and when they particles with a negative charge. He called these
are brought together, they attract particles electrons. Later, between 1909 and
each other. This phenomenon occurs 1911, another scientist named Ernest Rutherford
because the upper sheet acquires discovered that atoms also have a heavy centre
a different charge from the lower called the nucleus, which has a positive charge.
sheet. Conversely, when the two Normally, an atom is neutral, which means it
plastic sheets are rubbed separately is not charged, because the positive charge in
and then brought closer, they tend the nucleus and the negative charge from the
to repel one another due to both electrons are equal and cancel each other out.
sheets acquiring the same charge. But if the atom gets extra energy, it can lose
The process of charging through one of its outer electrons. When this happens,
rubbing involves the removal or the atom has more positive charge than negative
addition of electrons. For instance, charge, so it becomes positively charged. The
when the plastic sheets are rubbed electron that was removed can stay by itself or
against a sweater, electrons transfer join another atom. If it joins another atom, that
from the sweater to the upper plastic atom gets a more negative charge and becomes
3
Physics Form 2 Final.indd 3 25/10/2025 10:25