Page 10 - Physics_Form_2
P. 10
Physics for Secondary Schools
negatively charged. So, gaining or losing electric charge is really about moving electrons
from one atom to another. The charge on the electron equals 1.6 10 19 C. The proton
has a charge of the same magnitude as that of the electron but with an opposite charge.
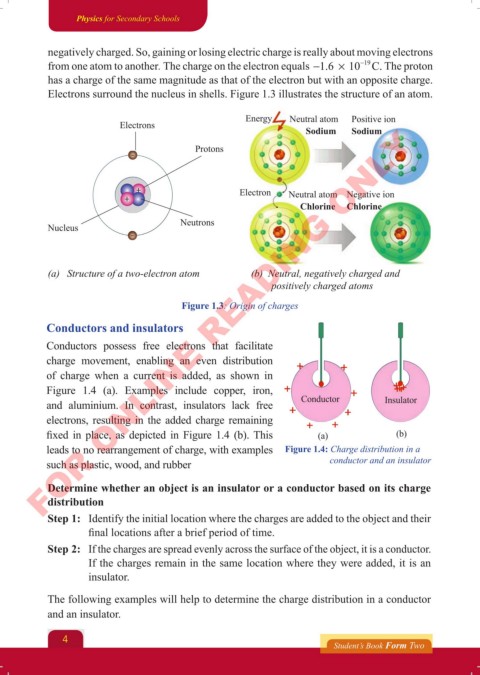
Electrons surround the nucleus in shells. Figure 1.3 illustrates the structure of an atom.
Energy Neutral atom Positive ion
Electrons
FOR ONLINE READING ONLY
Sodium Sodium
Protons
Electron Neutral atom Negative ion
Chlorine Chlorine
Neutrons
Nucleus
(b) Neutral, negatively charged and
(a) Structure of a two-electron atom
positively charged atoms
Figure 1.3: Origin of charges
Conductors and insulators
Conductors possess free electrons that facilitate
charge movement, enabling an even distribution
of charge when a current is added, as shown in
Figure 1.4 (a). Examples include copper, iron,
and aluminium. In contrast, insulators lack free Conductor Insulator
electrons, resulting in the added charge remaining
fixed in place, as depicted in Figure 1.4 (b). This (a) (b)
leads to no rearrangement of charge, with examples Figure 1.4: Charge distribution in a
such as plastic, wood, and rubber conductor and an insulator
Determine whether an object is an insulator or a conductor based on its charge
distribution
Step 1: Identify the initial location where the charges are added to the object and their
final locations after a brief period of time.
Step 2: If the charges are spread evenly across the surface of the object, it is a conductor.
If the charges remain in the same location where they were added, it is an
insulator.
The following examples will help to determine the charge distribution in a conductor
and an insulator.
4
Student’s Book Form Two
Physics Form 2 Final.indd 4 25/10/2025 10:25