Page 143 - Chemistry_Form_2
P. 143
Chemistry
for Secondary Schools
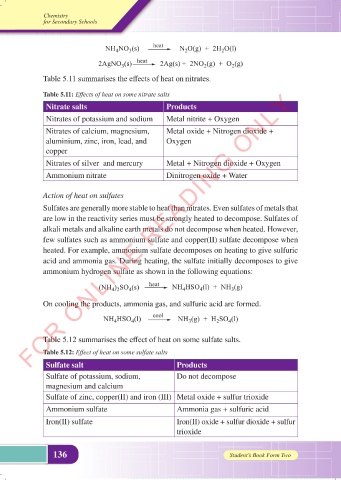
Table 5.11 summarises the effects of heat on nitrates.
FOR ONLINE READING ONLY
Table 5.11: Effects of heat on some nitrate salts
Nitrate salts Products
Nitrates of potassium and sodium Metal nitrite + Oxygen
Nitrates of calcium, magnesium, Metal oxide + Nitrogen dioxide +
aluminium, zinc, iron, lead, and Oxygen
copper
Nitrates of silver and mercury Metal + Nitrogen dioxide + Oxygen
Ammonium nitrate Dinitrogen oxide + Water
Action of heat on sulfates
Sulfates are generally more stable to heat than nitrates. Even sulfates of metals that
are low in the reactivity series must be strongly heated to decompose. Sulfates of
alkali metals and alkaline earth metals do not decompose when heated. However,
few sulfates such as ammonium sulfate and copper(II) sulfate decompose when
heated. For example, ammonium sulfate decomposes on heating to give sulfuric
acid and ammonia gas. During heating, the sulfate initially decomposes to give
ammonium hydrogen sulfate as shown in the following equations:
On cooling the products, ammonia gas, and sulfuric acid are formed.
Table 5.12 summarises the effect of heat on some sulfate salts.
Table 5.12: Effect of heat on some sulfate salts
Sulfate salt Products
Sulfate of potassium, sodium, Do not decompose
magnesium and calcium
Sulfate of zinc, copper(II) and iron (III) Metal oxide + sulfur trioxide
Ammonium sulfate Ammonia gas + sulfuric acid
Iron(II) sulfate Iron(II) oxide + sulfur dioxide + sulfur
trioxide
136 Student’s Book Form Two
01/08/2025 11:21:11
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 136 01/08/2025 11:21:11
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 136