Page 139 - Chemistry_Form_2
P. 139
Chemistry
for Secondary Schools
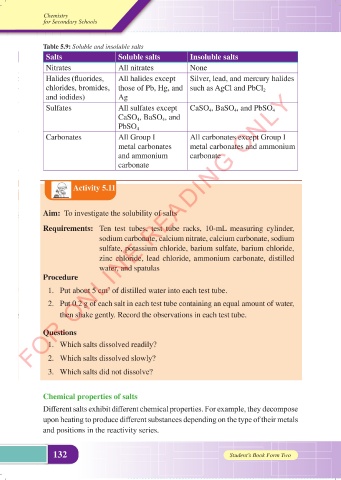
Table 5.9: Soluble and insoluble salts
Salts Soluble salts Insoluble salts
Nitrates All nitrates None
Halides (fluorides, All halides except Silver, lead, and mercury halides
FOR ONLINE READING ONLY
chlorides, bromides, those of Pb, Hg, and such as AgCl and PbCl 2
and iodides) Ag
Sulfates All sulfates except CaSO 4 , BaSO 4 , and PbSO 4
CaSO 4 , BaSO 4 , and
PbSO 4
Carbonates All Group I All carbonates except Group I
metal carbonates metal carbonates and ammonium
and ammonium carbonate
carbonate
Activity 5.11
Aim: To investigate the solubility of salts
Requirements: Ten test tubes, test tube racks, 10-mL measuring cylinder,
sodium carbonate, calcium nitrate, calcium carbonate, sodium
sulfate, potassium chloride, barium sulfate, barium chloride,
zinc chloride, lead chloride, ammonium carbonate, distilled
water, and spatulas
Procedure
1. Put about 5 cm of distilled water into each test tube.
3
2. Put 0.2 g of each salt in each test tube containing an equal amount of water,
then shake gently. Record the observations in each test tube.
Questions
1. Which salts dissolved readily?
2. Which salts dissolved slowly?
3. Which salts did not dissolve?
Chemical properties of salts
Different salts exhibit different chemical properties. For example, they decompose
upon heating to produce different substances depending on the type of their metals
and positions in the reactivity series.
132 Student’s Book Form Two
01/08/2025 11:21:10
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 132 01/08/2025 11:21:10
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 132