Page 54 - Chemistry_Form_2
P. 54
Chemistry
for Secondary Schools
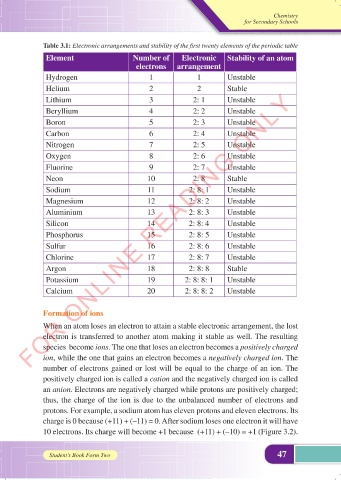
Table 3.1: Electronic arrangements and stability of the first twenty elements of the periodic table
Element Number of Electronic Stability of an atom
electrons arrangement
Hydrogen 1 1 Unstable
Helium 2 2 Stable
FOR ONLINE READING ONLY
Lithium 3 2: 1 Unstable
Beryllium 4 2: 2 Unstable
Boron 5 2: 3 Unstable
Carbon 6 2: 4 Unstable
Nitrogen 7 2: 5 Unstable
Oxygen 8 2: 6 Unstable
Fluorine 9 2: 7 Unstable
Neon 10 2: 8 Stable
Sodium 11 2: 8: 1 Unstable
Magnesium 12 2: 8: 2 Unstable
Aluminium 13 2: 8: 3 Unstable
Silicon 14 2: 8: 4 Unstable
Phosphorus 15 2: 8: 5 Unstable
Sulfur 16 2: 8: 6 Unstable
Chlorine 17 2: 8: 7 Unstable
Argon 18 2: 8: 8 Stable
Potassium 19 2: 8: 8: 1 Unstable
Calcium 20 2: 8: 8: 2 Unstable
Formation of ions
When an atom loses an electron to attain a stable electronic arrangement, the lost
electron is transferred to another atom making it stable as well. The resulting
species become ions. The one that loses an electron becomes a positively charged
ion, while the one that gains an electron becomes a negatively charged ion. The
number of electrons gained or lost will be equal to the charge of an ion. The
positively charged ion is called a cation and the negatively charged ion is called
an anion. Electrons are negatively charged while protons are positively charged;
thus, the charge of the ion is due to the unbalanced number of electrons and
protons. For example, a sodium atom has eleven protons and eleven electrons. Its
charge is 0 because (+11) + (–11) = 0. After sodium loses one electron it will have
10 electrons. Its charge will become +1 because (+11) + (–10) = +1 (Figure 3.2).
Student’s Book Form Two 47
01/08/2025 11:20:54
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 47 01/08/2025 11:20:54
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 47