Page 57 - Chemistry_Form_2
P. 57
Chemistry
for Secondary Schools
(e) They are generally insoluble in non-polar solvents such as carbon
tetrachloride and hexane. Non-polar solvents contain bonds between atoms
with similar electronegativities.
(f) They are typically hard but brittle.
FOR ONLINE READING ONLY
(g) They have high densities.
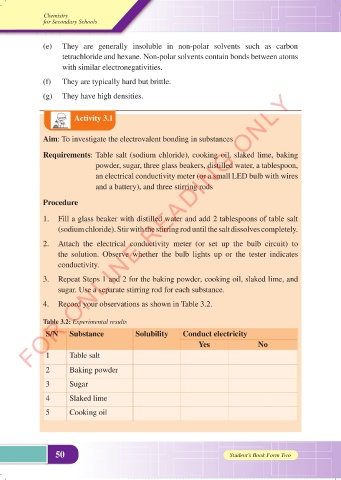
Activity 3.1
Aim: To investigate the electrovalent bonding in substances
Requirements: Table salt (sodium chloride), cooking oil, slaked lime, baking
powder, sugar, three glass beakers, distilled water, a tablespoon,
an electrical conductivity meter (or a small LED bulb with wires
and a battery), and three stirring rods
Procedure
1. Fill a glass beaker with distilled water and add 2 tablespoons of table salt
(sodium chloride). Stir with the stirring rod until the salt dissolves completely.
2. Attach the electrical conductivity meter (or set up the bulb circuit) to
the solution. Observe whether the bulb lights up or the tester indicates
conductivity.
3. Repeat Steps 1 and 2 for the baking powder, cooking oil, slaked lime, and
sugar. Use a separate stirring rod for each substance.
4. Record your observations as shown in Table 3.2.
Table 3.2: Experimental results
S/N Substance Solubility Conduct electricity
Yes No
1 Table salt
2 Baking powder
3 Sugar
4 Slaked lime
5 Cooking oil
50 Student’s Book Form Two
01/08/2025 11:20:55
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 50 01/08/2025 11:20:55
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 50