Page 115 - Chemistry_Form_2
P. 115
Chemistry
for Secondary Schools
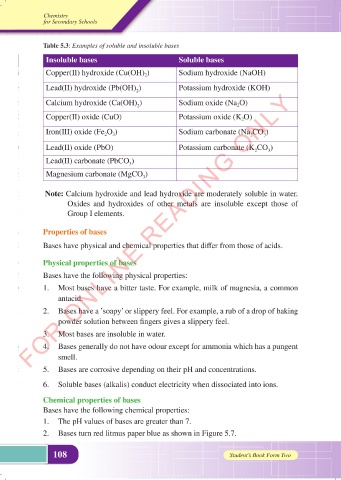
Table 5.3: Examples of soluble and insoluble bases
Insoluble bases Soluble bases
Copper(II) hydroxide (Cu(OH) 2 ) Sodium hydroxide (NaOH)
Lead(II) hydroxide (Pb(OH) ) Potassium hydroxide (KOH)
FOR ONLINE READING ONLY
2
Calcium hydroxide (Ca(OH) ) Sodium oxide (Na 2 O)
2
Copper(II) oxide (CuO) Potassium oxide (K 2 O)
Iron(III) oxide (Fe 2 O 3 ) Sodium carbonate (Na CO )
2
3
Lead(II) oxide (PbO) Potassium carbonate (K CO )
3
2
Lead(II) carbonate (PbCO )
3
Magnesium carbonate (MgCO )
3
Note: Calcium hydroxide and lead hydroxide are moderately soluble in water.
Oxides and hydroxides of other metals are insoluble except those of
Group I elements.
Properties of bases
Bases have physical and chemical properties that differ from those of acids.
Physical properties of bases
Bases have the following physical properties:
1. Most bases have a bitter taste. For example, milk of magnesia, a common
antacid.
2. Bases have a ’soapy’ or slippery feel. For example, a rub of a drop of baking
powder solution between fingers gives a slippery feel.
3. Most bases are insoluble in water.
4. Bases generally do not have odour except for ammonia which has a pungent
smell.
5. Bases are corrosive depending on their pH and concentrations.
6. Soluble bases (alkalis) conduct electricity when dissociated into ions.
Chemical properties of bases
Bases have the following chemical properties:
1. The pH values of bases are greater than 7.
2. Bases turn red litmus paper blue as shown in Figure 5.7.
108 Student’s Book Form Two
01/08/2025 11:21:03
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 108 01/08/2025 11:21:03
CHEMISTRY FORM TWO NEW 2025 DUMMY.indd 108